If your peptide supplier hands you a single number — “98% purity” — and calls it a day, you’re working with about a quarter of the picture.
Peptide quality is not one measurement. It’s a four-axis problem: how pure is it, what’s actually in it, how much is in the vial, and what came along for the ride from the synthesis lab. Skip any axis and you’re either dosing wrong, dosing the wrong molecule, or dosing the right molecule alongside a counter-ion that’s quietly skewing your results.
Here’s the analytical toolkit a serious peptide purity testing program should include — and how to read each piece without an analytical chemistry degree.
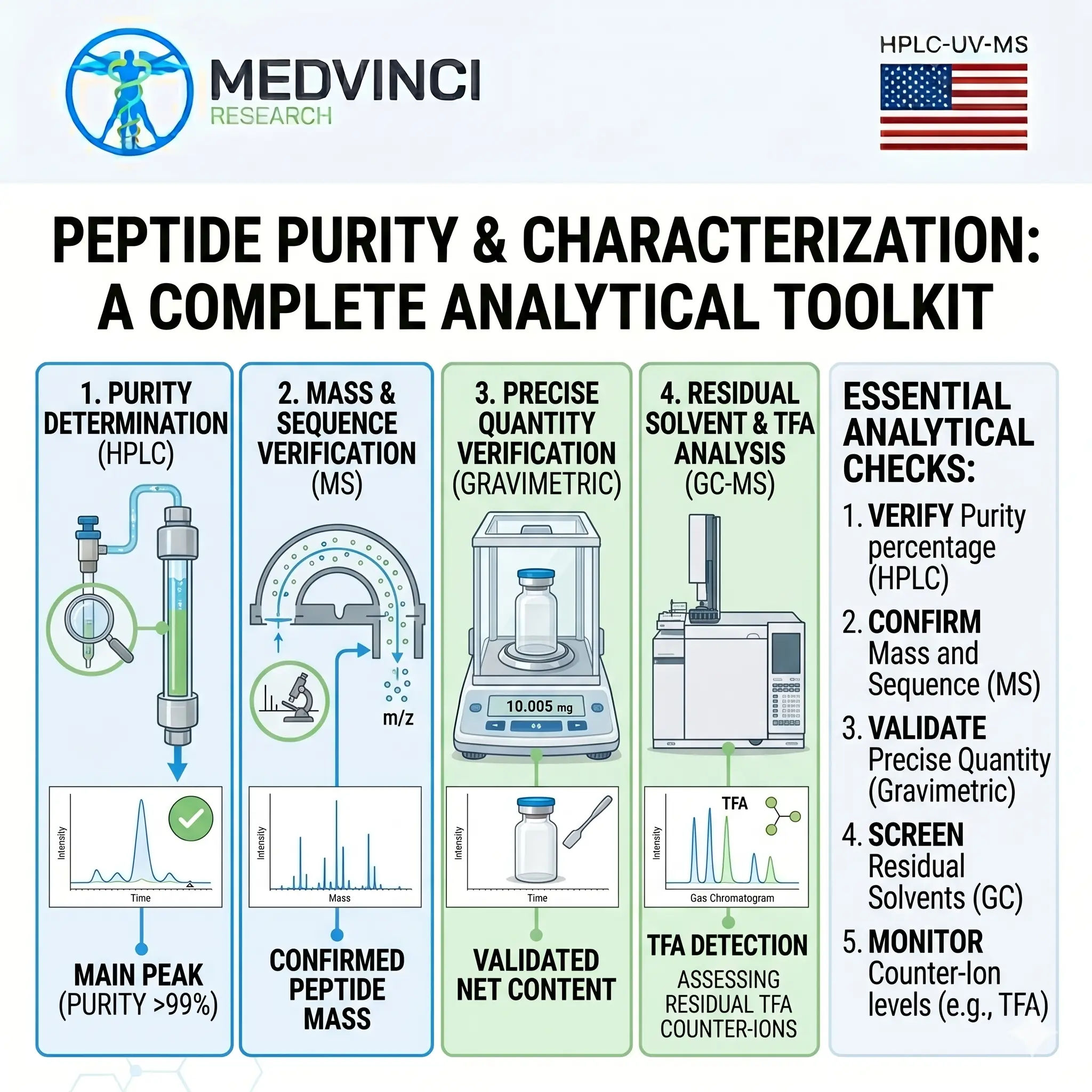
The four-test toolkit at a glance
- HPLC — purity (% target peptide vs. peptide-related impurities).
- Mass spectrometry — identity (is this actually the sequence you ordered?).
- Gravimetric assay — quantity (how many milligrams of peptide are really in the vial?).
- GC-MS — what came with it (residual solvents and counter-ion content).
Why peptide purity actually matters
Two reasons.
Reproducibility. If your peptide is 92% target and 8% deletion sequences, the second batch is unlikely to behave like the first. The impurity profile drifts lot-to-lot, and so does whatever biological signal you’re trying to measure.
Dosing accuracy. A 10 mg vial that’s 85% net peptide content — after subtracting water, salts, and counter-ions — is an 8.5 mg vial. If your protocol calls for 10 mg, you’re under-dosing by 15%. That’s the difference between a clean dose-response curve and a noisy one you can’t explain.
The four tests below address these problems from different angles. You want all four.
Test 1: HPLC — Purity Determination
What it measures: The percentage of the target peptide relative to all other peptide-related species in the sample.
How it works: High-Performance Liquid Chromatography (HPLC) separates the mixture by pushing it through a column that retains different molecules for different durations. UV detection — typically at 214 nm for the peptide bond, or 280 nm for aromatic residues — produces a chromatogram where the area under each peak represents the relative abundance of each species.
What “good” looks like: A single dominant peak integrating to ≥98% of total peak area. Research-grade peptides commonly target ≥95%; pharmaceutical-grade targets ≥99%.
Watch the wavelength
Suppliers reporting purity without specifying the wavelength, gradient, or detection method are skipping the part that matters. 98% pure at 254 nm is not the same as 98% at 214 nm. The 214 nm number is the honest one for peptides because almost every amino acid absorbs there. Higher wavelengths only see aromatic residues and quietly hide everything else.
Test 2: Mass Spectrometry — Mass & Sequence Verification
What it measures: The molecular mass of the peptide, which confirms you actually got the sequence you ordered.
How it works: Mass spectrometry (usually ESI-MS or MALDI-TOF for peptides) ionizes the molecule and measures its mass-to-charge ratio (m/z). The observed mass should match the theoretical monoisotopic or average mass calculated from the sequence — typically within 0.1 Da on a high-resolution instrument.
Why it matters: HPLC tells you the sample is pure. Mass spec tells you what it’s pure of. You can have a 99% pure sample of the wrong peptide. This happens more than the industry likes to admit, particularly with sequences that differ by a single amino acid substitution or a missing residue near the N-terminus.
Common mistake
A COA that shows HPLC but no MS spectrum is half a quality story dressed up as a whole one. Identity is not optional.
Test 3: Gravimetric Analysis — Precise Quantity Verification
What it measures: The actual mass of material in the vial, verified against the label.
How it works: Analytical balances accurate to 0.01 mg confirm net content. This is the least glamorous test on the list and the one most likely to bite you.
Why it matters: Lyophilized peptides almost always contain residual water, counter-ions (TFA or acetate), and trace salts. A vial labeled “10 mg peptide” may contain 10 mg of material, of which only 8.2 mg is actual peptide. Without a peptide content assay — sometimes called net peptide content or peptide assay by amino acid analysis (AAA) — you don’t know your real concentration.
Watch for the gross-vs-net distinction. Premium suppliers report both gross mass and net peptide content. Most report neither and let you assume.
Test 4: GC-MS — Residual Solvents & TFA Analysis
What it measures: Trace solvents left over from synthesis and counter-ion content — notably trifluoroacetic acid (TFA).
How it works: Gas Chromatography coupled with Mass Spectrometry (GC-MS) separates and identifies volatile compounds. For peptides, the targets are residual solvents (DMF, DCM, acetonitrile, methanol, isopropanol) and TFA, which is the default counter-ion left behind from reverse-phase HPLC purification.
Why TFA matters: TFA can account for 5–20% of the mass of a lyophilized peptide. It’s not biologically inert — it has documented effects on mitochondrial function and can confound results in sensitive in-vitro systems. For applications where TFA interference is a concern, peptides should be exchanged into acetate or HCl salts, and the new counter-ion content reverified.
Why residual solvents matter: Synthesis and purification solvents have ICH Q3C limits for a reason. Class 2 solvents like DCM and DMF are not what you want in a cell-culture experiment. For the regulatory context behind these limits, see our companion guide on the 2026 peptide regulatory framework.
The most common COA gap
COAs that omit residual solvent and counter-ion data entirely are the single most common gap in commercial peptide documentation. If you don’t see GC-MS, ask why.
The 5-point checklist for evaluating any peptide COA
Before you trust a vial, the certificate of analysis should clear all five:
- Verify purity percentage by HPLC — with method conditions stated (column, gradient, detection wavelength).
- Confirm mass and sequence by MS — with observed vs. theoretical mass shown.
- Validate precise quantity gravimetrically, ideally with both gross mass and net peptide content.
- Screen residual solvents by GC against ICH Q3C limits.
- Monitor counter-ion levels (typically TFA), with explicit % w/w reported.
Any COA missing two or more of these is incomplete. Any supplier who can’t answer follow-up questions about their analytical methods is selling you a number, not a measurement. For a deeper read on what a real peptide COA contains and how to spot the fakes, see How to Read a Peptide COA and Third-Party vs. Vendor-Only COAs.
Why most suppliers skip half of this
The honest answer: full analytical characterization costs money. A complete HPLC + MS + GC-MS + gravimetric workup runs roughly $150–$400 per batch depending on the contract lab. At low margins or high volume, suppliers cut GC-MS first, then the net peptide content assay, then MS, until what’s left is a single HPLC trace and a label.
That math works for the supplier’s P&L. It does not work for the researcher whose data depends on knowing what’s actually in the vial.
What this means for buyers
You don’t need to run the tests yourself. You need to insist on seeing them done.
When evaluating a peptide source, three questions cut through almost any sales pitch:
- Can you send me a representative COA with HPLC, MS, GC-MS, and net peptide content?
- What’s your typical TFA % w/w?
- What detection wavelength is your HPLC purity reported at?
A supplier who answers all three without flinching has built the analytical infrastructure. A supplier who deflects, redirects, or sends a one-page summary with no method details is asking you to take their word for it. Your data is too expensive to make on faith.
Frequently asked questions about peptide purity testing
What’s the difference between peptide purity and peptide content?
Purity is the percentage of target peptide relative to other peptide-related impurities (deletion sequences, oxidized variants, truncations). Content is the percentage of the vial mass that is actually peptide, after subtracting water, salts, and counter-ions. A peptide can be 99% pure and only 80% content — and both numbers can be true on the same COA.
Is 95% purity good enough for research peptides?
For most in-vitro and exploratory animal work, yes — provided the impurity profile is characterized and consistent batch-to-batch. For receptor binding studies, structure-activity work, or anything dose-sensitive, 98%+ is the working standard.
Why is there TFA in my peptide?
TFA is the standard mobile-phase acid used in reverse-phase HPLC purification. It binds to basic residues (Lys, Arg, His) as a counter-ion and is co-lyophilized with the peptide unless explicitly exchanged into a different salt form.
Do I need GC-MS data on every batch?
For routine work with a validated supplier, periodic GC-MS verification is acceptable. For new suppliers, new sequences, or any batch destined for a sensitive assay, request it on the specific lot you’re using.
What does “≥99% by HPLC at 214 nm” actually mean?
It means that when the sample was run on a reversed-phase HPLC column and detected at 214 nm (the peptide bond absorbance), the target peak accounted for at least 99% of the total integrated peak area. It does not address non-peptide impurities — salts, solvents, water, counter-ions. That’s what the other three tests are for.
What’s the difference between MALDI-TOF and ESI-MS for peptides?
Both confirm peptide mass. MALDI-TOF is faster and tolerates more sample contamination, making it the workhorse for routine confirmation. ESI-MS gives higher mass accuracy and couples cleanly to LC for impurity identification. For QC release, either is acceptable; for impurity characterization, ESI-MS is the better tool.
How do I know if a COA is real?
Real COAs include: lot number, manufacture date, retest date, the actual chromatograms and spectra (not just summary numbers), method parameters, and a signature or QC release stamp. A one-page table of numbers with no underlying data is a certificate of compliance, not a certificate of analysis. They are not the same document. The Medvinci public COA directory shows what a third-party-verifiable batch record actually looks like.
The bottom line
Peptide purity is a single line on a four-line story. The other three lines — identity, quantity, and what came with it — are where the experimental noise actually lives.
Demand all four. The suppliers who can deliver them will respect you for asking. The ones who can’t have just told you something useful.
Not industry standard. Should be.
Medvinci Research publishes a complete analytical package — HPLC purity, MS mass and sequence verification, gravimetric net content, and GC-MS residual solvent and TFA analysis — with every batch. Browse the public COA directory to see real batch records, or open a research account to track every batch you receive.



